
In this shell, electrons are placed one at a time, beginning from the top position and going clockwise. 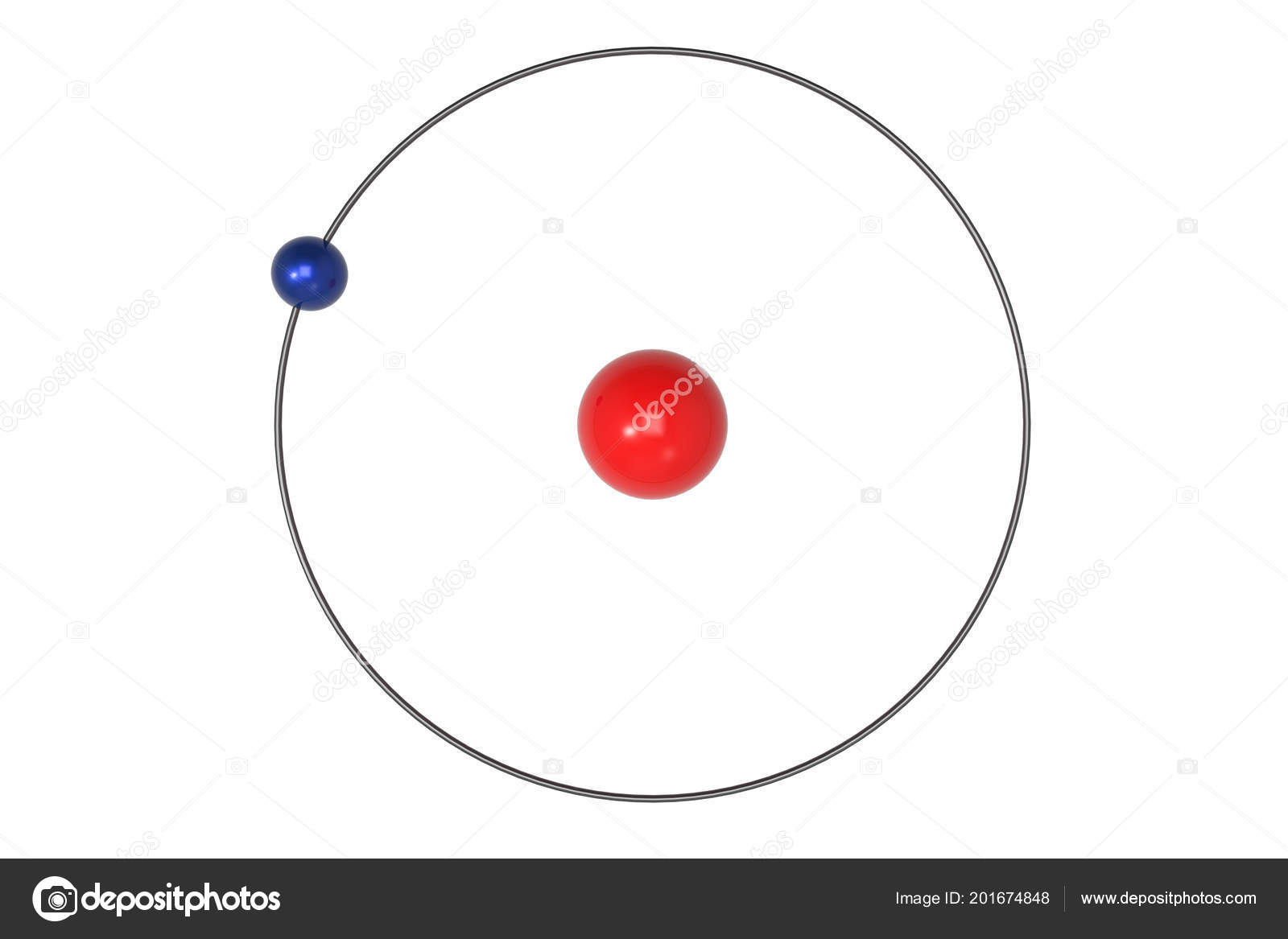
So, place the remaining 3 electrons in the L shell. The boron atom has 5 electrons, so place two electrons from it in the K shell. Step 3:ĭraw a circle around the nucleus, i.e., the first orbital shell K. Sketch the nucleus, which is a small circle made up of the number of protons and neutrons. Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. Boron has an atomic number of 5 and an atomic mass of 10.811, which means it has 5 protons, 5 electrons, and 11 neutrons. Step 1:Ĭount the protons, electrons, and neutrons. To understand how to draw the bohr model of boron, let’s go through the steps with this helpful diagram. The outermost shell holds 3 valence electrons which are essential for understanding the chemistry of Boron. Its nucleus contains 6 neutrons and 5 protons, ringed by two electron shells named K-shell and L-shell. Let us understand this in detail with the example of the Bohr Atomic Model of Boron.īoron is a fascinating element. To move from lower to higher energy levels, electrons gain energy, while they lose energy on moving from higher to lower energy levels. The first orbital shell, n=1, is assigned as K, n=2 as L, n=3 as M, and so on.Įlectrons in the lowest energy level, n=1, are considered to be in the ground state. These orbital shells are identified by an integer, n, also known as quantum numbers. It proposed that the electrons revolve around the nucleus of an atom in well-defined circular paths, known as orbits or shells.Įach orbit or shell has a fixed energy and is known as the orbital shell or energy level. 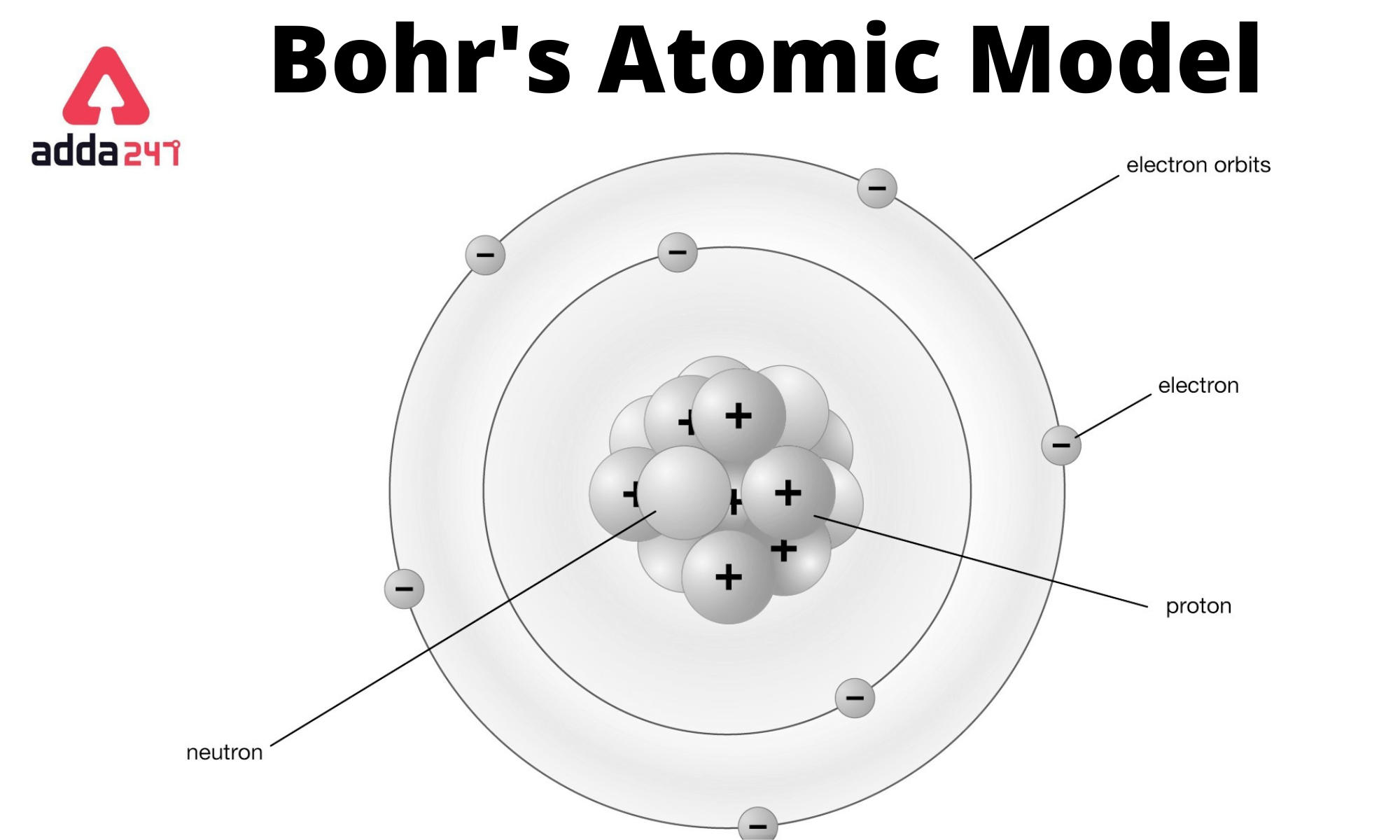
This work’s success lies in its ability to justify Rydberg’s formula for hydrogen spectral series, which was never experimentally confirmed before. It is one of the most important scientific discoveries, as it laid the groundwork for further breakthroughs in understanding the atomic structure and its implications for physics and chemistry. The Bohr model is just like the planets orbiting around the sun in the solar system. The Bohr Model, developed by Niels Bohr in 1913, revolutionized our understanding of atomic structure. So, according to Niels Bohr, let’s start understanding the mysterious world of atoms. Let’s clarify things and show you how important this model is for scientists.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
